Associação Portuguesa de Investigação em Cancro
Introducing innate immunity to 3D tumour models
Introducing innate immunity to 3D tumour models
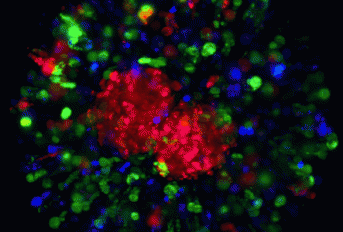
iBET researchers developed a new 3D cell culture model to study the impact of the immune microenvironment of solid tumours in tumour progression and therapeutic response. Cancer drug discovery has been limited by high rates of failure in clinical trials, despite positive laboratory tests suggesting good drug activity. Several types of tumour cell models are used in research labs and by the pharmaceutical industry for discovery and pre-clinical testing of new anti-cancer drugs. Nevertheless, these models still fail to reconstruct the complexity of human solid tumours, as they usually do not account for the role of the immune system in tumour progression and response to therapy. Researchers from the Animal Cell Technology Unit – Advanced Cell models Lab have developed a cell model employing tumour and stromal cells, as well as macrophages. The researchers showed that the model recapitulates features of the tumour niche, including the crosstalk between the different cell players that leads to the immunosuppressive and invasive microenvironment typical of advanced stage carcinomas. In this innovative model, macrophages can infiltrate the tumour mass and acquire a tumour-promoting phenotype. By challenging the model with an immunotherapeutic agent, the authors demonstrated its applicability in testing the efficacy of novel immune-targeting agents, as well as its suitability to study in further detail the molecular mechanisms involved in that response. These results have been recently published in Biomaterials, one of the top leading journals in biotechnology. “We have developed a cell model in which the immunosuppressive microenvironment of advanced stage tumours can be recapitulated. Moreover, its design facilitates the adaptation to industrial scales, and therefore can be a useful tool in cancer drug discovery”, state Sofia Rebelo and Catarina Pinto, the co-first authors of the study. “Our model is a new tool to study human macrophage plasticity within the tumoral context and to address tumour-immune interactions in response to external stimuli, such as chemotherapeutic and immunomodulatory drugs”, refers Catarina Brito, head of the Advanced Cell Models Lab (http://www.itqb.unl.pt/research/technology/advanced-cell-models/advanced...) from the Animal Cell Technology Unit (http://tca.itqb.unl.pt), the leader of this study. This work was carried out at iBET (www.ibet.pt) and ITQB NOVA (http://www.itqb.unl.pt/) and within the scope of the IMI-funded project PREDECT (http://www.predect.eu/about/) and of iNOVA4Health (http://www.inova4health.com/), in close collaboration with academic partners (IPOLFG, Lisbon; ICFO, Barcelona) and a pharmaceutical company (Boehringer Ingelheim, a member of the PREDECT consortium). The published paper is freely accessible to all.
Rebelo, S. P. (1, 2), Pinto, C. (1, 2), Martins, T. R. (1, 2), Harrer, N. (3), Estrada, M. F.(1, 2), Loza-Alvarez, P. (4), Cabeçadas, J. (5), Alves, P. M. (1, 2), Gualda, E. J. (4), Sommergruber, W. (3), Brito, C.(1, 2)
(1) iBET, Instituto de Biologia Experimental e Tecnológica, Apartado 12, 2780-901, Oeiras, Portugal
(2) Instituto de Tecnologia Química e Biológica António Xavier, Universidade Nova de Lisboa, Av. da República, 2780-157, Oeiras, Portugal
(3) Boehringer Ingelheim RCV GmbH & Co KG, Department of Lead Discovery, 1121, Vienna, Austria
(4) ICFO - Institut de Ciències Fotòniques, The Barcelona Institute of Science and Technology, 08860, Castelldefels, Barcelona, Spain
(5) Serviço de Anatomia Patológica, Instituto Português de Oncologia de Lisboa Francisco Gentil, Lisboa, Portugal
The tumour microenvironment (TME) shapes disease progression and influences therapeutic response. Most aggressive solid tumours have high levels of myeloid cell infiltration, namely tumour associated macrophages (TAM). Recapitulation of the interaction between the different cellular players of the TME, along with the extracellular matrix (ECM), is critical for understanding the mechanisms underlying disease progression. This particularly holds true for prediction of therapeutic response(s) to standard therapies and interrogation of efficacy of TME-targeting agents. In this work, we explored a culture platform based on alginate microencapsulation and stirred culture systems to develop the 3D-3-culture, which entails the co-culture of tumour cell spheroids of non-small cell lung carcinoma (NSCLC), cancer associated fibroblasts (CAF) and monocytes. We demonstrate that the 3D-3-culture recreates an invasive and immunosuppressive TME, with accumulation of cytokines/chemokines (IL4, IL10, IL13, CCL22, CCL24, CXCL1), ECM elements (collagen type I, IV and fibronectin) and matrix metalloproteinases (MMP1/9), supporting cell migration and promoting cell-cell interactions within the alginate microcapsules. Importantly, we show that both the monocytic cell line THP-1 and peripheral blood-derived monocytes infiltrate the tumour tissue and transpolarize into an M2-like macrophage phenotype expressing CD68, CD163 and CD206, resembling the TAM phenotype in NSCLC. The 3D-3-culture was challenged with chemo- and immunotherapeutic agents and the response to therapy was assessed in each cellular component. Specifically, the macrophage phenotype was modulated upon treatment with the CSF1R inhibitor BLZ945, resulting in a decrease of the M2-like macrophages. In conclusion, the crosstalk between the ECM and tumour, stromal and immune cells in microencapsulated 3D-3-culture promotes the activation of monocytes into TAM, mimicking aggressive tumour stages. The 3D-3-culture constitutes a novel tool to study tumour-immune interaction and macrophage plasticity in response to external stimuli, such as chemotherapeutic and immunomodulatory drugs.
Biomaterials
https://doi.org/10.1016/j.biomaterials.2018.02.030






